Special Access Medical Device
Legal/Guidance Documents:
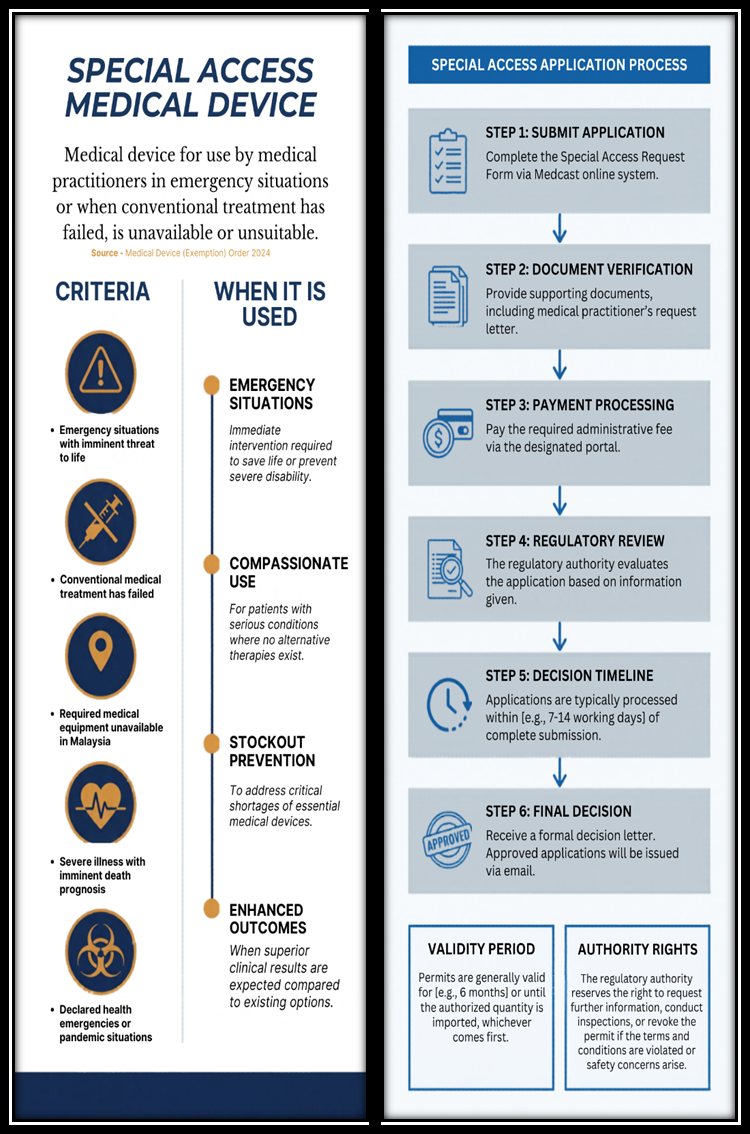
How to Submit Application :
- Application form : Medcast
- How to create Medcast Account : Medcast – Notification Creation
- Administrative Fee : RM 300
Reference Documents:
- Essential Requirement: Medcast form
- Medical Practitioner’s Request Letter : Template
- Post handling : Disposal Form
Contact Information:
- General Email: [ sa.cm@mda.gov.my ]
- Admin : Mohamad Aznil | ☎ +603-82300 0247
Officers in Charge :
- Pn. Nur Maizura Bt Zarmani | ☎ +603-8230 0339
- Pn. Aidahwaty Ariffin @ M.Olaybal, A.M.N | ☎ +603-8230 0364
Important Notice :
1. Please be advised that applications that do not meet stated criteria or lack proper justification will be rejected.
2. To qualify for Special Access Notification, all applicants must possess a valid establishment license, specifically as an Authorized Representative and Importer.
Prepared by: Pre Market Control Division
Uploaded by: Corporate Communication Division
Updated on: 11 May 2026






