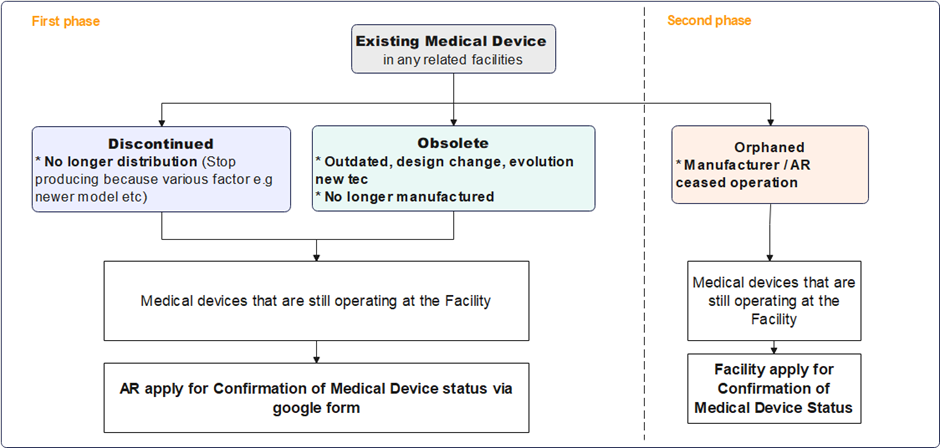
Obsolete, Discontinued & Orphaned Medical Device
Full Implementation of Notification for Orphaned, Obsolete, and Discontinued Medical Devices
Update :
The Medical Device Authority (MDA) would like to provide an update on the implementation timeline for the Notification of Orphaned, Obsolete, and Discontinued (OOD) Medical Devices. This initiative, originally set to launch in full by November 2024, will now be introduced in two phases to ensure comprehensive guidance and smooth implementation:
- Phase 1: Discontinued and Obsolete (OD) Medical Devices
The guidance document for OD medical devices is in the final stages of completion, and the application for exemption process for these categories will commence following its release.
- Phase 2: Orphaned Medical Devices
The development of a separate guidance document specifically for orphaned medical devices is underway. The application for exemption process for orphaned devices will begin upon completion of this additional guidance document.
Suggestion Framework (Updated version, will be notified):
Any inquiries, please email to : ood.md@mda.gov.my
Updated: 30th Oct 2024
Personal Use Medical Device
A. Introduction
Importation and placement of a medical device in the Malaysian market requires the medical device to comply with the requirements of the Medical Device Act 2012 (Act 737), and the medical device shall be registered with the Medical Device Authority (MDA) under Section 5 of Act 737. However, the Medical Device (Exemption) Order 2024 has provided an exemption from medical device registration and establishment license requirements for importation of personal use medical devices.
B. Scope of Exemption
The exemption applicable to personal use medical devices under the Medical Device (Exemption) Order 2024 are as follows:
1. Exemption from registration of medical devices
According to the Medical Device (Exemption) Order 2024, medical devices for the purpose of personal use have been exempted from the requirement for registration of medical devices under Section 5 of Act 737.
2. Exemption from establishment license
A person who imports medical devices for the purpose of personal use is exempted from the requirement of an establishment license under Section 15 of Act 737.
C. Definition of Personal Use Medical Device
A medical device which is brought into Malaysia for the use of a particular individual only and not to be placed in the market or be used by a third party.
[SOURCE: Medical Device (Exemption) Order 2024
NOTE: Refers to Section 43 in the Act 737 for explanation on a third party.
D. Requirements for Importing or Purchasing Personal Use Medical Devices
1. Medical devices for personal use may be imported or purchased subject to the following requirements:
a) The medical device shall be for the use of immediate family, and not for commercial purposes, resale, and distribution.
NOTE: Personal use medical devices must be purchased by an individual (under their own name) and cannot be purchased by an establishment or organization
b) To provide a formal prescription or letter of recommendation on the professional use medical device(s) from a registered healthcare professional, upon request from the Authority.
NOTE: Certain types of medical devices may not require recommendation from healthcare professionals.
Refer Annex A for examples.
c) for medical devices shall be appropriate according to the type of the medical device and there are a few examples as specified in Attachment 1 as reference.
NOTE:
Low/medium risk medical device (eg: Glucose Monitoring, Hearing Aids, In-vitro diagnostic products) : 1 piece of each type.
Consumable medical device (eg: contact lens, In-vitro maintenance test strips ) : Quantity for 3 months supply.
d) all labels and labelling information that comes with the medical device shall be retained; and
e) no same or similar medical device(s) is/are registered in Malaysia
NOTE: Please refer to Medical Device Authority Register (MDAR) : https://mdar.mda.gov.my
2. The importation of medical devices for the purpose of personal use is also exempted from the requirements of import permit under Customs (Prohibitions of Imports) Order 2023.
E. Regulatory Framework for Importation of Personal Use Medical Device
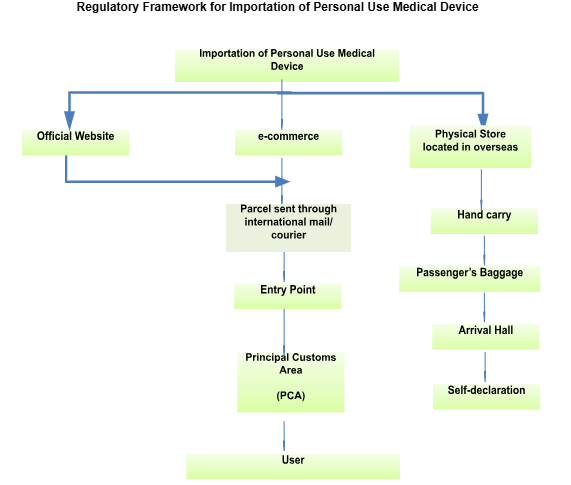
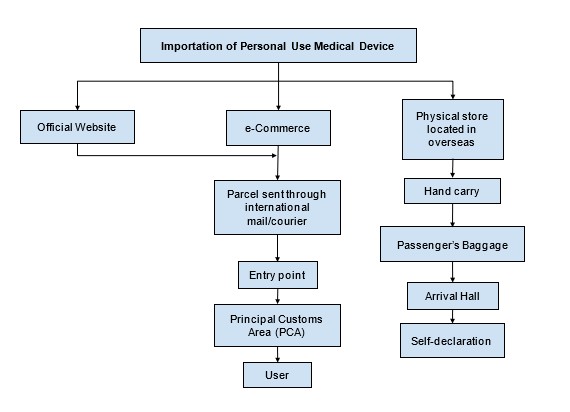
Any disputes or inquiries can be submitted via email to exemption.bhai@mda.gov.my or contact our representative, Pn. Nur Aisyah at 03 8230 0253
F. EXAMPLES AND SITUATIONS: Examples and Situations of Personal Use Medical Device
Date of upload: 22 December 2025
Prepared by: Policy and Strategic Planning Division
Uploaded by: Corporate Communication Division
List of Device Studies (Device Study Tracker)
![]()
Please click link to view list of device studies tracker as below:
https://docs.google.com/spreadsheets/d/1BR9xYVYDdvv2mi0QO7pp1r4NIeyHn18Qv3mf0AhylSI/edit?usp=sharing
Updated: 29th March 2023
Special Access Medical Device
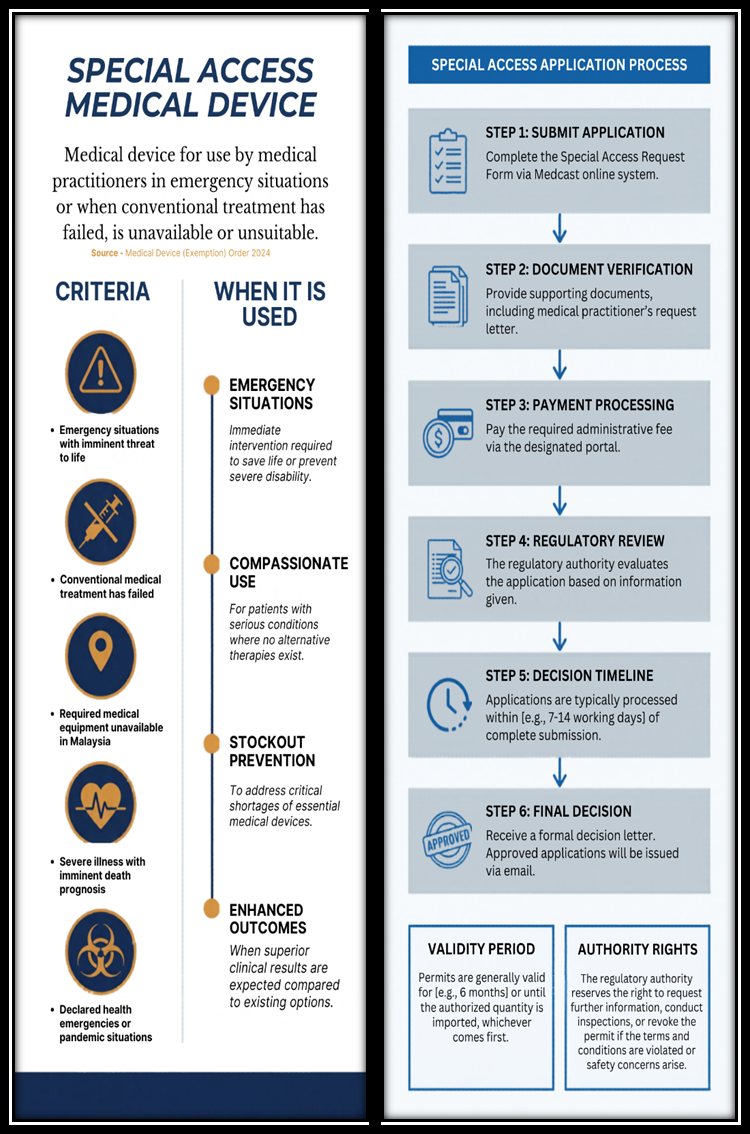
Legal/Guidance Documents:
How to Submit Application :
- Application form : Medcast
- How to create Medcast Account : Medcast – Notification Creation
- Administrative Fee : RM 300
Reference Documents:
- Essential Requirement: Medcast form
- Medical Practitioner’s Request Letter : Template
- Post handling : Disposal Form
Contact Information:
- General Email: [ sa.cm@mda.gov.my ]
- Admin : Mohamad Aznil | ☎ +603-82300 0247
Officers in Charge :
- Pn. Nur Maizura Bt Zarmani | ☎ +603-8230 0339
- Pn. Aidahwaty Ariffin @ M.Olaybal, A.M.N | ☎ +603-8230 0364
Important Notice :
1. Please be advised that applications that do not meet stated criteria or lack proper justification will be rejected.
2. To qualify for Special Access Notification, all applicants must possess a valid establishment license, specifically as an Authorized Representative and Importer.
Prepared by: Pre Market Control Division
Uploaded by: Corporate Communication Division
Updated on: 11 May 2026
Custom-made Medical Device
|
Announcement: Clarification on the regulation of Dental Products The Medical Device Authority (MDA) wishes to clarify that dental products, including but not limited to crowns and aligners, are medical devices that fall under the regulatory requirements of the Medical Device Act 2012 [Act 737]. Accordingly, all local companies including laboratories involved in the manufacturing of such dental products must ensure that these devices are registered with the Authority to comply with Act 737. In cases where the dental products meet the criteria for custom-made medical devices, manufacturers may submit an exemption application directly to the Authority. This exemption is applicable only when the specific requirements of a custom-made device are fulfilled. This announcement serves to reinforce the importance of regulatory compliance and to support the safe and effective use of dental products in Malaysia. Date of published: 11-Nov-2024 |
Custom made Criteria :
1. Designed and manufactured according to a written prescription from a qualified medical practitioner for the sole use of a particular patient.
2. Does not fall into the category of mass-produced medical devices that require adaptation for specific professional user requirements.
** Medcast form requirement: - Crucial to adhere (click here) -
How to apply?
A. Notification form : Medcast
B. How to create Medcast Account : Medcast – Notification Creation
C. Administrative Charge : RM 300
D. Guidance Documents : NOTIFICATION OF CUSTOM-MADE (MDA/GD/0064)
E. Any inquiries, please email to sa.cm@mda.gov.my
F. Contact No. relevant Officer:
- Puan Nur Maizura Bt Zarmani +603-8230 0339
- En. Mohamad Aznil bin Ahmad Azmi (Printing Officer) +603-8230 0247
Process flow:
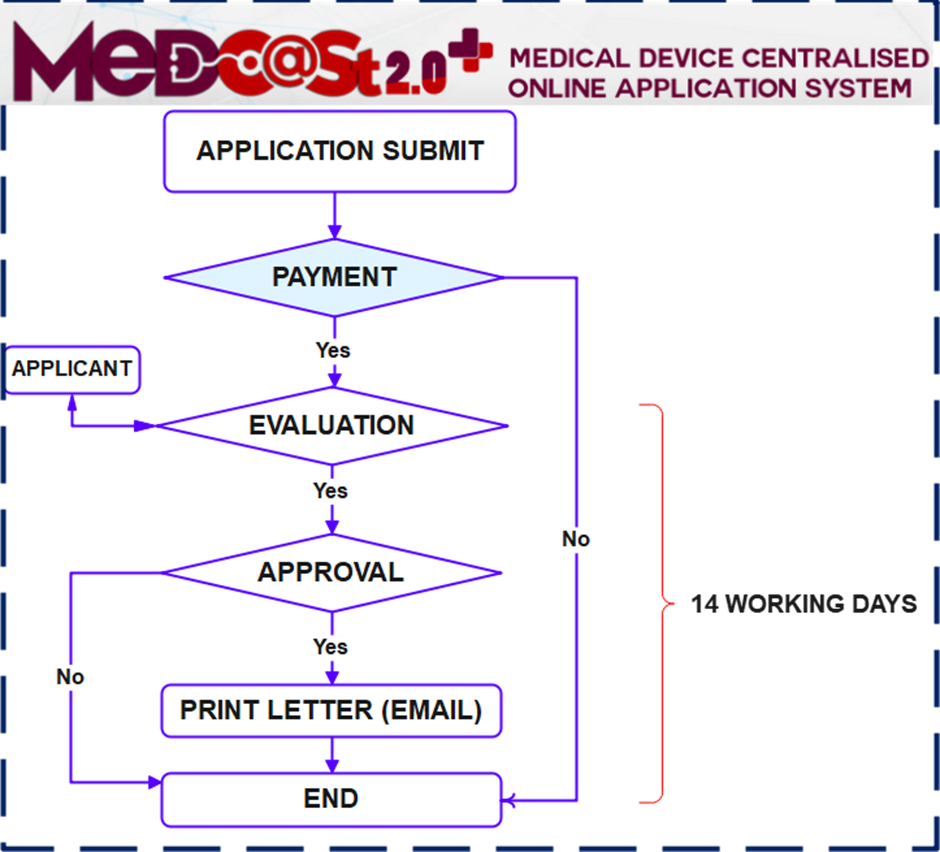
Importance Notice :
- Kindly note that commencing 1 December 2023, all submissions for Custom Made Medical Device Notifications are required to be made through the MeDC@St 2.0 online platform. The application processing period is 14 working days upon receipt of a complete application.
- The Custom-Made Notification System is activated using earlier version of Medcast 2.0. Users may face system bugs, but rest assured, it won't impact your application's submission. Applicants are advised to refer the Medcast Form Requirement to prevent any potential issues. If you encounter unresolved issues, please reach out to the MDA officer for assistance.
Updated: 8th October 2025






