ADVERTISEMENT OF MEDICAL DEVICE
What is an Advertisement?
 Legal Framework
Legal Framework
Section 44 (1): No person shall advertise a medical device unless the medical device is registered and complies with the Act.
Section 44 (2): Advertisements must not make any misleading or fraudulent claims.
Medical Device (Advertising) Regulations 2019
No person shall advertise a registered medical device without approval from the Authority.
How to Submit a Medical Device Advertisement Application?
To submit an advertisement application for medical devices, please refer to the following guidelines and resources that clarify the requirements for advertisements that require or do not require approval:
Reference Documents:
- Guidance Documents MDA/GD/0032: Code of Advertisement – Outlines the standards and principles for advertising medical devices.
- Guidelines: Application for Medical Device Advertisement Approval - Requirements – Details the approval process and necessary documentation.
Application Form:
- Advertisement Application Online Form – Digital submission platform for advertisement approval.
- Template for an Advertisement Letter of Authorisation – Standard template for authorisation letters related to advertisement applications.
The process flow for an advertisement application is as follows:
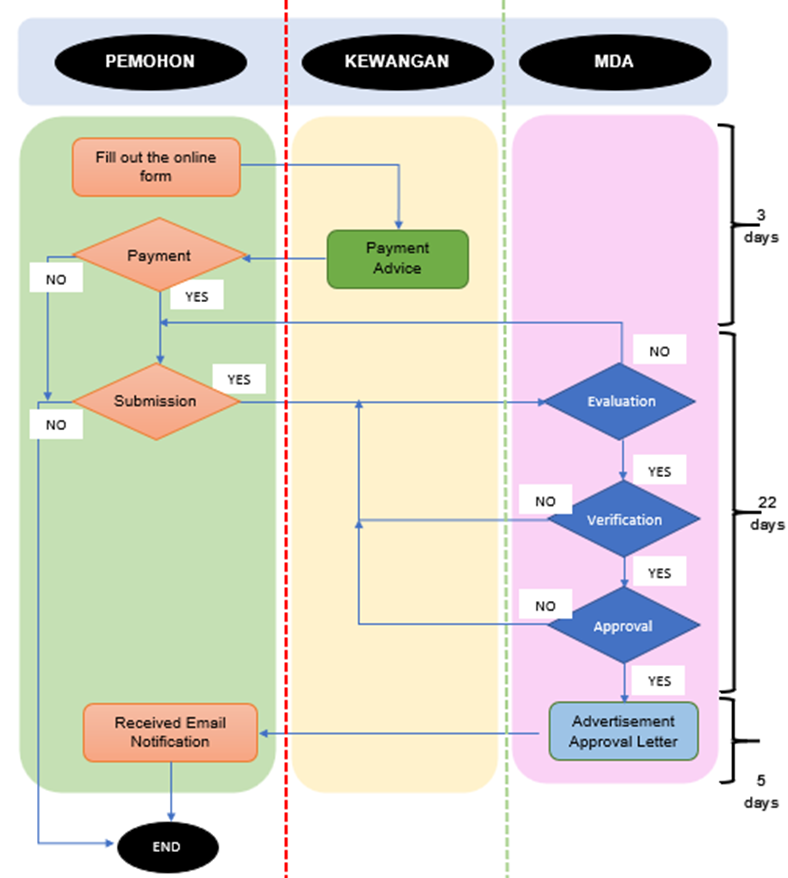
|
IMPORTANCE NOTICE: 1. Claims Must Be Accurate and Aligned with Registration All advertising claims must strictly align with the product’s approved registration. Claims must be consistent with the intended use, fully supported by documentation. Any advertisement containing exaggerated, unsupported, or unapproved claims will be rejected. 2. Prohibited Claims and Content All advertisement claims must be developed in accordance with the Code of Advertisement (COA). |
Contact Information
Advertisement Application Process (e.g. application submission) :
- General Email: [ advertisment@mda.gov.my ]
- Administration : Mohamad Aznil | Tel: + (03-82300 0247)
Officers in Charge :
- Hasdiana Mohammadiah | ☎ +603-8230 0364 | ? hasdiana@mda.gov.my
- Nur Maizura Zarmani | ☎ +603-8230 0339 | ? maizura@mda.gov.my
- Dinesh Murgis | ☎ +603-8230 0393 | ? dineshmurgis@mda.gov.my
- Zahroh Hasanah Darwis | ☎ +603-8230 0388
Prepared by: Advertisement Unit ( Pre Market Control Division)
Uploaded by: Corporate Communication Division
Date of upload: 5 February 2026
NOTIFICATION FOR EXPORT ONLY MEDICAL DEVICE
Notification of Export Only Medical Device
1. Introduction
- The importation, exportation, or placement of a medical device in the Malaysia market requires the medical device to be registered under Medical Device Act 2012 (Act 737). However, under Medical Device (Exemption) Order 2024 state that medical devices for purpose of export only are exempted from registration requirements and shall make an application for an exemption to the Authority.
- The exportation of unregistered medical device is allowed once the Export Only Medical Device Exemption Letter is issued by the Authority.
2. Requirements for Notification of Export Only Medical Device
- Applicant shall submit a notification before exportation of the first shipment.
- The application Export Only Medical Device Exemption shall be submitted via google form and the required documents are as follows:
|
No |
Documents
|
|
1 |
Registrar of Companies (ROC) certificate of applicant |
|
2 |
|
|
3 |
|
|
4 |
(Please refer to Figure 2: Determination whether LOA is required or not for Export Only Exemption) |
|
5 |
A copy manufacturer’s QMS ISO 13485 certificate |
|
6 |
A copy of Brochure/Leaflet/Label/IFU that contain information on brief description and intended use. |
|
7 |
A copy of establishment license (If applicable) |
Note: *new requirements for Notification of Export Only Medical Device
3. Determination whether LOA is required or not for Notification of Export Only Medical Device?
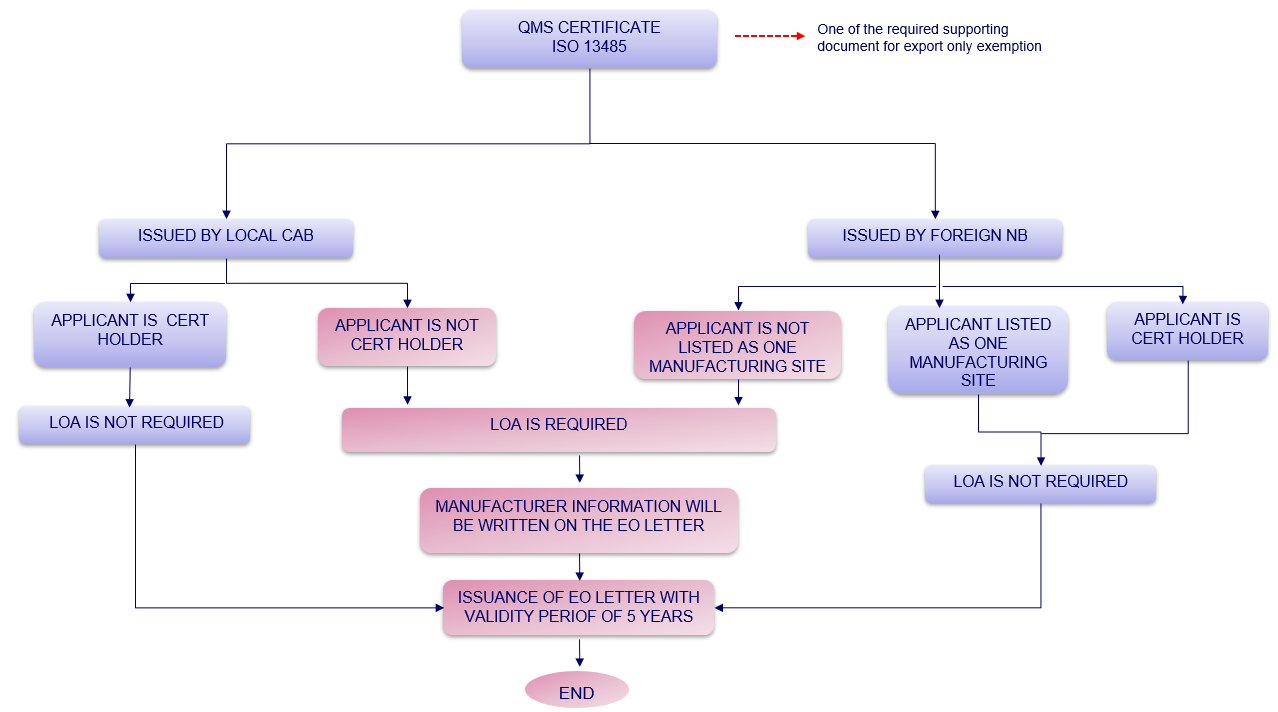
Figure 1: Determination whether LOA is required or not for Notification of Export Only Medical Device
4. Notification of Export Only Medical Device Process
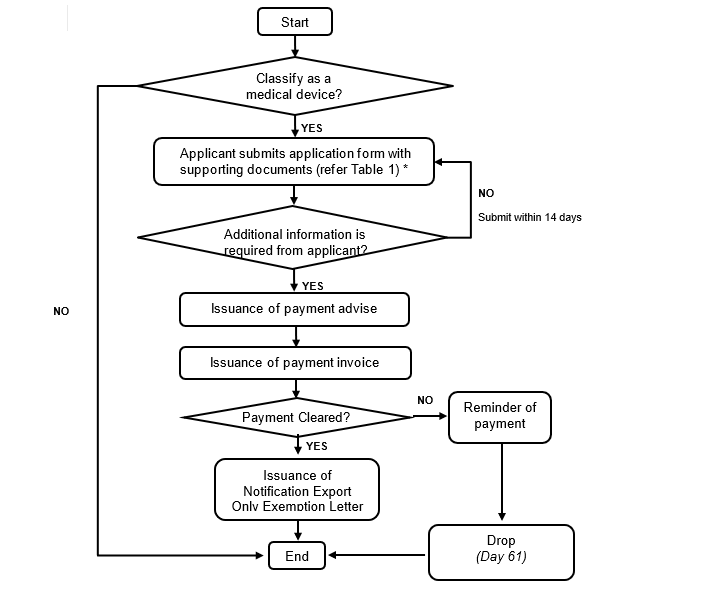
Figure 2: Notification of Export Only Medical Device Process
5. Fees for Notification of Export Only Medical Device
The payment RM500 shall be made online via portal BayarNow. For guidance on online payment is accessible via link or qr code below:

6. Issuance of Export Only Medical Device Exemption Letter
- Upon receipt of completed application and clearance of payment, the authority will issue an Export Only Medical Device Exemption Letter to the applicant within 14 days after clearance of payment, by letter and email.
- This Export Only Medical Device Exemption Letter permits multiple export consignments within the validity period of the letter.
- The validity period of the Export Only Medical Device Exemption Letter is five (5) years
- Once Export Only Medical Device Exemption Letter has expired or has been cancelled/withdrawn, no further export of the medical device, at any quantity, shall be permitted.
Any inquiries please email to exportonly.ec@mda.gov.my
General Line: +603 8230 0300
Direct Line : +603 8230 0253 (Pn. Nur Aisyah)
+603 8230 0208 (Pn. Hafizah)
+603 8230 0299
Updated: 21 / 04 / 2026
List of User Manual and Guidance Document
|
No |
Title |
Download |
|
1. |
User Manual: i.Establishment Licence Application ii.Renewal of Establishment Licence Application iii. Amendment Major/Minor Application iv. Withdrawal Application |
|
|
2. |
User Manual for Change Of Ownership Application |
|
|
3. |
User Manual Video of Establishment Licence Application |
|
|
4. |
User Manual Video for Renewal of Establishment Licence Application |
|
|
5. |
User Manual Video of Amendment Major/Minor Application |
|
|
6. |
User Manual Video of Withdrawal Application |
|
|
7. |
User Manual Video of Change Of Ownership Application |
|
|
8. |
Guidance Document of Licensing for Establishment |
|
|
9. |
Guidance Document for Good Distribution Practice for Medical Devices (GDPMD) |
|
|
10. |
Guidance Document for Change Of Ownership Application |
|
Consultation
ANNOUNCEMENT ON MDA CONSULTANCY SERVICES
MDA offers regulatory consulting that can support stakeholders who are involve in regulatory submission for medical devices in Malaysia, to comply with medical device regulatory requirements under Medical Device Act 2012 (Act 737). The regulatory consulting services include:
Online consultancy maybe requested for applicant who isn’t physically able to meet MDA’s consultancy team. Consultancy *fee imposed is based on type of consultancy services selected.
*Note: Please refer to Consultancy Request Form (Package) and Consultancy Request Form (Non-Package)
The following diagram describes the step-by-step process of requesting our consultancy services:
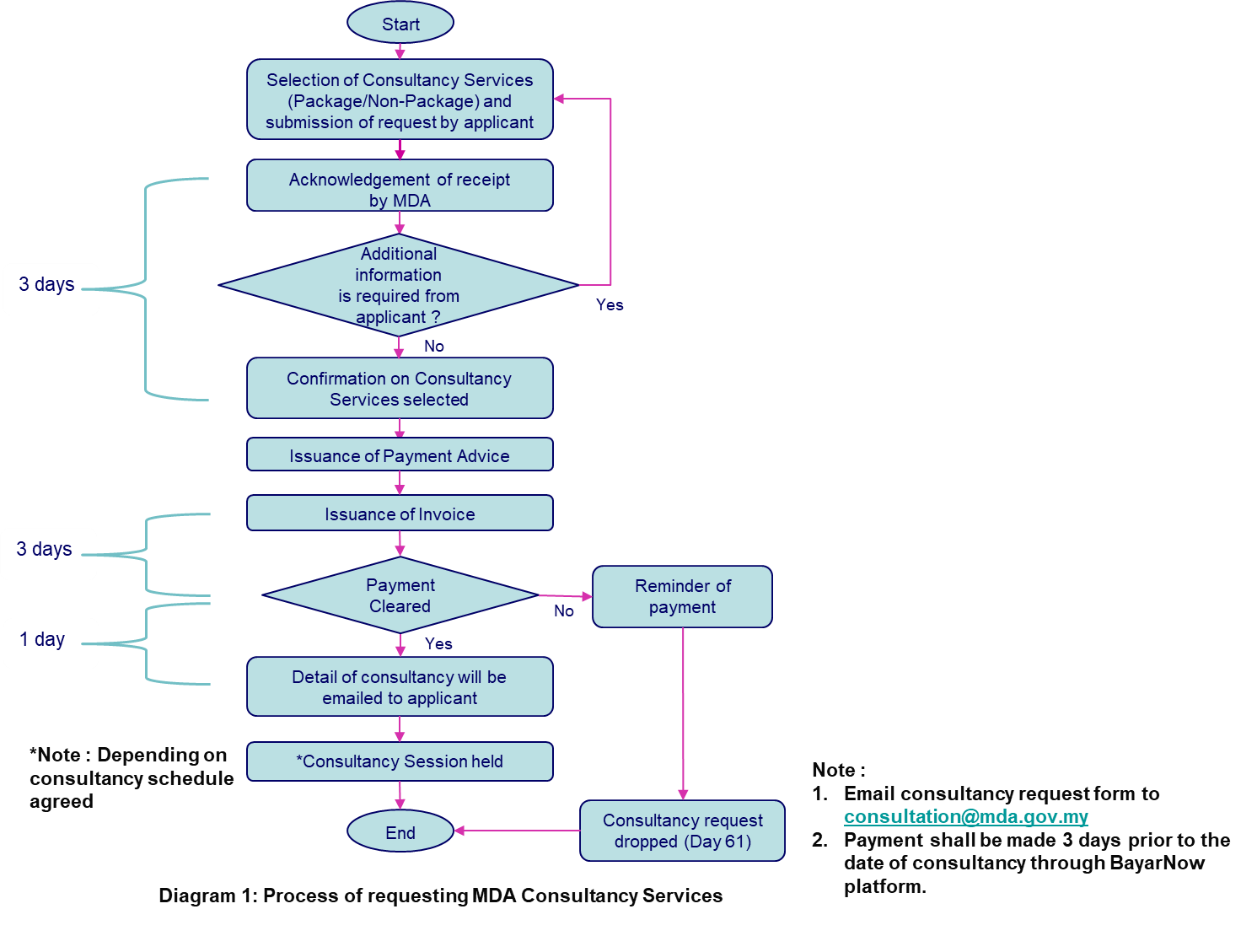
If you want to know more about our consultancy services, send an email to consultation@mda.gov.my
Updated: 21 August 2023
Certificate Free Sale / Manufacturing Certificate
Local companies wishing to apply for Certificate of Free Sale (CFS) / Manufacturing Certificate (MC), please CLICK HERE for online application.
ANNOUNCEMENT:
Conditions and fee structure:
- One CFS/MC application is only for 25 medical device registration numbers. If more than 25 registration number, please apply in another separately CFS/MC application form. This is not applicable to the non-registered medical device.
- All medical devices under 1 application must be from same manufacturer. If the manufacturer are different, please apply in different CFS/MC application form
- All applications will be charged RM 100 per registration number per country for 2 years validity; and all applications will be charged RM 300 per registration number per country for 5 years validity.
- Certificate is inclusive of 1 attachment. Additional pages will be charged RM5.00 per page. Each attachment will list up to 25 medical devices.
- Certificate will be issued based on per country.
- Font type Arial and size 10 shall be used for attachments.
Step for Application:
- Section 1 is for applicant details;
- Section 2 is for details of manufacturer;
- Please fill the document on product list ( CLICK HERE ) and upload into section for document to be uploaded. Please upload only 1 attachment of product list in 1 application
- Required documents to be uploaded:
|
Documents |
Certificate of free sale |
Manufacturing Certificate (export only manufacturer) |
Manufacturing Certificate (OEM manufacturer) |
|
Registrar of Companies (ROC) certificate of applicant |
Yes |
Yes |
Yes |
|
A copy of establishment license |
Yes |
Yes |
Not applicable |
|
Proof of medical device registration (Notification letter / Acknowledgement letter/ Registration certificate) |
Yes |
Yes |
Not applicable |
|
Yes |
Yes |
Yes |
|
|
Letter of declaration as OEM |
Not applicable |
Not applicable |
Yes |
|
Brochure |
No |
No |
Yes |
(And any other additional information, particulars /documents as may be required)
- Once status is checked in CFS online application system. Payment invoice will be uploaded in the BayarNow system in 3 working days;
- Certificate issuance will be emailed after payment is cleared by Finance Unit MDA after 7 working days.
PAYMENT:
- All fees shall be paid via BayarNow system. CASH and BANK DRAFT WILL NOT BE accepted. We will not be responsible for the cash sent or BANK DRAFT brought to MDA;
- For instructions on how to use BayarNow, please download the user manual at the following link, MDA BayarNow User Manual.
Additional information
Additional information, particulars and documents requested by the Authority on the application shall be submitted by the applicant within 14 working days from the date of return of application. If the additional information, particulars or document of the application is not given by the applicant within the time granted (14 working days), the application shall be deemed to be withdrawn and shall not be further proceed with, but without affecting the right of the applicant to make a fresh application.
Complete submission for certificate of free sales application will be processed within 14 working days
The above requirement is subject to change from time to time.
Any enquiries can be sent to Medical Device Authority (MDA) at the following address:
Chief Executive
Medical Device Authority (MDA), Ministry of Health Malaysia
Level 6, Prima 9, Prima Avenue II,
Block 3547, Persiaran APEC, 63000 Cyberjaya,
Selangor, MALAYSIA
Tel: +603-8230 0300 | Fax : +063-8230 0200
Or, please contact:
1) Mr Fezri Aziz +03-8230 0395
Email: cfs@mda.gov.my
2) Mr Fadhli Zil +03-82300357
Email: cfs@mda.gov.my
Updated: 27 October 2025
Prepared by: International & Industry Relations Division
Uploaded by: Corporate Communication Division






